Stars of Research
We’re involved in more than 300 projects looking to find new treatments and advances in patient care, covering many areas of medicine and with thousands of patients volunteering to take part in clinical trials. Here are some of our Stars of Research – who you can also follow by using #StarsofResearch. Nominate a Star of Research yourself by emailing Communications@nnuh.nhs.uk.
NNUH joins inflammatory bowel disease (IBD) studies
 Two new studies have been launched at our hospital to help further enhance clinical understanding of inflammatory bowel disease (IBD) amongst children and young people.
Two new studies have been launched at our hospital to help further enhance clinical understanding of inflammatory bowel disease (IBD) amongst children and young people.
More than 100 children and 3,000 adults are currently under the care of the hospital for IBD and three to four new cases a month are being diagnosed by the paediatric gastroenterology team. IBD covers conditions such as ulcerative colitis and Crohn’s disease, which can be debilitating for children and young adults as well as affecting their mental health and wellbeing.
The Paediatric Gastroenterology team and Children’s and Women’s Research team at NNUH have joined the TRIPP Research Protocol (Translational Research in Intestinal Physiology and Pathology) in Cambridge, which is funded and supported by the National Institute for Health and Care Research (NIHR). The study involves collecting blood samples and intestinal biopsies to understand the genetic signatures that can cause IBD. Mini-gut models can also be grown from stem cells to further understanding of the mechanisms involved in IBD and to test specific treatments.
Vaccination halves risk of long Covid, new study shows
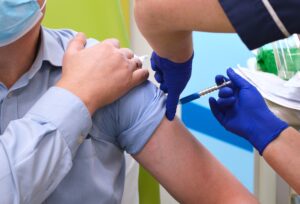 Being vaccinated against Covid halves people’s risk of developing long Covid, according to new research from our Trust and the University of East Anglia.
Being vaccinated against Covid halves people’s risk of developing long Covid, according to new research from our Trust and the University of East Anglia.
Long Covid affects around two million people in the UK, and new research, published in JAMA Internal Medicine, reveals the risk factors associated with developing the condition.
Overweight people, women, smokers and those over the age of 40 are also more likely to suffer from long Covid according to the study – which includes more than 860,000 patients and is thought to be the largest of its kind.
The study also finds that co-morbidities such as asthma, COPD, Type 2 Diabetes, coronary heart disease, immunosuppression, anxiety and depression are also associated with increased risk of long Covid.
Orthopaedic research published in top medical journal
Older knee replacement designs are just as effective as newer models – according to new research from our orthopaedic research team and University of East Anglia.
A new study published in the journal BMJ Open compares the effectiveness of two established knee replacements. Eighty osteoarthritis patients, who received total knee replacements in 2018 and 2019, took part the CAPAbility study – a blinded randomised controlled trial run by researchers at NNUH and UEA.
NNUH diabetes research published in top journal

Intermittently scanned glucose monitoring technology significantly improves the management of Type 1 diabetes, according to research conducted in Norfolk and published in a prestigious health research journal.
The findings of a study conducted at the Norfolk and Norwich University Hospital and seven other Trusts across the country has been published in the New England Journal of Medicine, which makes the case for more patients to have access to continuous blood glucose monitoring technology.
A total of 156 participants took part in the Flash UK trial, which was funded by Diabetes UK, with half monitoring their own blood glucose levels with finger prick testing and half using intermittently scanned continuous glucose monitoring over a six-month period.
The research at NNUH was led by Dr Sankalpa Neupane, Consultant in Diabetes and Endocrinology at NNUH.
Theatre team take part in research to help older patients

Our anaesthetists are taking part in a new national research project aimed at improving the care of older patients having surgery.
A team of doctors at NNUH are taking part in the Royal College of Anaesthetists-run SNAP3 project, which will run for the next two weeks.
The study is investigating the effects of frailty on outcomes in surgery and will look at both the long-term and short-term effects, including postoperative delirium, an uncommon but distressing condition which is much more likely to happen to older patients.
Anaesthetists take part in new obstetric research

A team of anaesthetists have been taking part in a new research study assessing the recovery of new mothers who have had a baby at our Trust.
An obstetric anaesthetic research team has recruited patients to the National Institute for Health Research (NIHR) supported ObsQoR study. A team of four anaesthetic registrars were involved in data collection and two Obstetric Anaesthetic Consultants who recruited 27 participants.
The research study assesses factors affecting the recovery of mothers who have had an anaesthetic intervention during labour or delivery. Our Trust is one of 70 centres participating across the UK in the study, which is sponsored by Guy’s and St Thomas’ NHS Foundation Trust.
The team consists of Consultant Anaesthetists Dr Joanna Walker (PI), Dr Rocio Ochoa-Ferraro and Anaesthetic trainees Dr Tinesh Mathew, Dr Lydia Rhodes, Dr Jon Dearden and Dr Harriette Beard.

Our Older People’s Medicine research team and UEA School of Pharmacy are working together with clinicians and researchers across the country on a new project called CHARMER (CompreHensive geriAtRician-led Medication Review) – a group of studies designed to develop an approach to increasing the chances of medicines that may cause harm or discomfort in the future being stopped when people are in hospital.

More than 500 participants from NHS, Norwich Research Park and local community are taking part in the Novavax Covid-19 vaccine trial in Norfolk, which in being run at the NNUH-run Clinical Research Facility at the Quadram Institute.
Norfolk Diabetes Prevention Study

Providing support to help people with prediabetes make small changes to their lifestyle, diet and physical activity can almost halve the risk of developing Type 2 diabetes, according the results of the Norfolk Diabetes Prevention Study (NDPS) – the largest diabetes prevention research study in the world in the last 30 years.
Wearable device research to help orthopaedic patients

Patients who have hip or knee replacements are set to get more support with their recovery following the launch of a new research study that uses wearable technology to monitor walking patterns.
The Norfolk and Norwich University Hospital (NNUH) has joined forces with Dynamic Metrics Ltd (DML) to develop and test a system to study mobility and improve a patient’s gait after a joint replacement.
International recognition for Norfolk radiologist

A Norfolk radiologist has achieved international recognition for his research using advanced imaging to help patients with osteoarthritis and other musculoskeletal conditions.
Dr Jamie MacKay, who is a radiologist at the Norfolk and Norwich University Hospital (NNUH) and lecturer at University of East Anglia (UEA), has been elected as a Junior Fellow of the International Society for Magnetic Resonance in Medicine (ISMRM), which has around 8,000 members.
Exploring the link between poor diet and post-stroke falls

Dietitians here and at the University of East Anglia are carrying out research aiming to help reduce the number of falls suffered by patients recovering from a stroke.
Our Research Capability Fund (RCF) has awarded £12,500 for a year-long project set up by dietitian Eliza Tassone to examine whether patients who suffer a stroke are more likely to have a subsequent fall when their nutritional status is poor.

We’re very proud to be involved in RECOVERY, a national study that’s testing existing drugs to see if they are effective in treating Covid-19.
The RECOVERY trial is a large, randomised controlled trial of possible treatments for patients admitted to hospital with Covid-19. More than 11,500 patients. The first significant results found that the use of the steroid dexamethasone cut the risk of death by a third for patients on ventilators and reduced deaths by a fifth for patients on oxygen.
Using a cannabis-based medicine on NICU

We’ve enrolled the world’s very first two babies onto this randomised study, which is funded and sponsored by GW Pharmaceuticals and supported by the National Institute for Health Research (NIHR).
Researchers say this is the first step that could one day lead to a cannabis-derived medicine being used routinely in neonatal care to help babies at risk of seizures and brain injury. The study is looking to see if the medicine is safe and effective in lessening the degree of brain injury for babies with Neonatal Hypoxic-ischemic Encephalopathy (HIE).
Boosting gut health of premature babies

Boosting milk for premature babies with healthy bacteria may have helped halve the number of serious gut problems and infections, according to new research we’ve led.
Researchers at here at NNUH, Quadram Institute and the University of East Anglia reviewed the outcomes of almost 1,000 very premature babies who were admitted to the Neonatal Intensive Care Unit over a 10-year period.
We recruited the highest number of people onto this nationwide research study across 40 hospitals and involving 472 patients.
MUK9 used state-of-the-art genetic profiling and a novel drug regime that’s tailored to an individual’s genetic subtype to treat high-risk bone marrow cancer.

Patients with chronic hand eczema have been taking part in a research study here to help establish the most effective treatment for their condition.
We’re one of the top recruiters in the country to the ALPHA trial and 20 of our patients have so far joined the study, co-ordinated by the Leeds Institute of Clinical Trials Research, which involves participants having their hands exposed to ultra violet light or taking the drug Alitretinoin.

The Norfolk Accumulation of Dietary Bioactives and Prostate Cancer (ADaPt) study was launched by researchers here and at Quadram Institute Bioscience (QIB).
Taking place at the NNUH-run Clinical Research Facility at the Quadram Institute, it involves 40 patients due to receive a biopsy for suspected or previously diagnosed prostate cancer, and is investigating the link between consuming the bioactive compounds in broccoli and garlic and prostate cancer to expand our understanding of how they can improve prostate health.
The Broccoli In Osteoarthritis (BRIO) trial

Researchers here and at the University of East Anglia have launched a trial to see whether eating broccoli could help with osteoarthritis.
They will investigate the compound sulforaphane, released when eating cruciferous vegetables like broccoli, Brussels sprouts and cabbage with trial participants who are over 50 and in pain with knee osteoarthritis – who like broccoli.
The Broccoli In Osteoarthritis (BRIO) trial will investigate whether a broccoli-rich diet improves pain and physical function. Researchers will compare the effects of eating broccoli soup with eating one that contains no broccoli but looks and tastes the same, measuring pain and physical function at the start of the trial, at six weeks and at 12 weeks.

We’re the first in the country to enrol patients on to the PEP-TALK trial, co-ordinated by the University of Oxford, which aims to help patients to be more active following surgery and improve their quality of life.
Physiotherapists, including Lewis Weatherburn and Helena Daniell (pictured) joined the study to provide more support to patients who have received hip or knee replacements.

We were the first Trust in the country to join the FROSTTIE clinical trial to help babies with breastfeeding difficulties because of tongue-tie – which affects up to 11% of newborns.
The study aims to discover if skilled support for breastfeeding on its own, or together with a minor snip (frenotomy), helps mothers and their babies to breastfeed.
Luisa Lyons, Specialist Midwife for Restrictive Lingual Frenulum and Lactation Support, and Louise Coke, Senior Paediatric Research Nurse, are working on the study.

We were the first hospital in the East of England to sign-up to the NeoCLEAR trial, which aims to improve the treatment of babies with suspected infections or neurological conditions.
Karen Few and Raducu Clapuci are working on the trial in our Neonatal Intensive Care Unit, investigating the best techniques to carry out a successful lumbar puncture procedure.
A lumbar puncture is performed by doctors to diagnose meningitis or other neurological conditions. The procedure involves taking a small amount of fluid from the spine through a needle in the lower back and the main aim of the clinical trial is to find out which technique produces the clearest sample of fluid in the lowest number of attempts.

Our Dermatology research team was first to sign-up a patient to the Sunshine study, which aims to help patients with the painful skin condition Hidradenitis suppurativa (HS). The condition leaves people requiring constant management and they often need surgery.
The clinical trial, sponsored by Novartis and supported by the NIHR (National Institute for Health Research), is being run across 33 countries.
LuCID – NNUH joins biopsy breathalyser trial

Eleanor Mishra, Consultant in Respiratory Medicine, and Julie Cook are supporting the LuCID biopsy breathalyser trial.
A lot of lung cancers are diagnosed late and the study is looking at early detection by measuring volatile organic compounds (VOCs) in exhaled breath.
Patients referred to us with suspected lung cancer took part in the LuCID trial by giving a breath sample which will help develop a potential screening programme. The study is led by Royal Papworth Hospital and was developed by Owlstone Medical, who created a biopsy breathalyser.

Our Oncology team, including Adele Cooper and Jenny Nobes, began the DANTE clinical trial to help patients who are receiving immunotherapy to treat inoperable melanoma.
While immunotherapy appears to be most effective in the first year, some patients with skin cancer have received the treatment for more than two years.
The new trial, funded by the National Institute for Health Research (NIHR), will find out whether stopping immunotherapy after a year is as effective at controlling the cancer as treatments over a longer period.

Principal Investigator John Phillips, Consultant Ear, Nose and Throat Surgeon, Jacob Newman and Stephen Cox hope that, following trials, the CAVA device will be available at initial referral to help treat dizziness.
Dizziness can be the result of problems with the inner ear or other causes, including heart and circulatory, neurological or metabolic (such as diabetes) conditions, and even anxiety. This means its often hard to identify the exact cause of sporadic attacks of dizziness in many patients. Currently, our CAVA device is unique and provides a special opportunity to gain insight into the workings of the ears and brain.

We do a lot of research to try to keep at the forefront of neonatal care. Everything we’re doing with our research involving babies is trying to work out what is better for babies and what treatments may help them to survive as healthily as possible.
We can only move forward if we do research and are grateful to all the parents who generously allow their babies to participate in these important research studies.


